Domains
Protein Domains
Protein domains are segments of a protein that serve a functional purpose. They are often important to the overall function of the protein and are generally conserved across a variety of species [1]. There are a variety of protein databases that can be used to identify key domains within a protein of interest. These include, but are not limited to, SMART, PROSITE, and Pfam. Identifying domains within proteins can help with determining a protein's function.
HFE Protein Domains
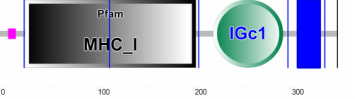
Two databases, SMART and Pfam, were used to identify the domains in the human HFE protein. Both databases were able to identify the immunoglobulin C1-set domain (IGc1) , but only Pfam was able to identify the Class I Histocompatibility Antigen domain (MHC_1). Only SMART was able to identify the transmembrane region shown in blue. The pink box in the SMART image indicates a low complexity region.
The IGc1 domain, which is involved in immune response, is commonly found alongside MHC_1 domains. Interestingly HFE has been classified as an atypical MHC protein [2]. So although HFE contains the MHC and IGc1 domains, it does not participate in immune response like other proteins with these domains would.
Two of the common HFE mutations, H63D and S65C, are found in the MHC_1 domain. The more severe mutation, C282Y, is found in the IGc1 domain.
Two of the common HFE mutations, H63D and S65C, are found in the MHC_1 domain. The more severe mutation, C282Y, is found in the IGc1 domain.
Human HFE Domain Analysis
The fact that the C282Y mutation, which is most commonly associated with hemochromatosis, is found in the IGc1 domain suggests that this domain is very important for its overall function in iron uptake. However, the H63D and S65C mutations also occur within a domain- MHC_1. Although these mutations need to be found in conjunction with a C282Y mutation and generally do not result in as severe of phenotypes as an individual with two copies of the C282Y mutation, this suggests that this domain is also somehow very important for HFE's role in regulating iron. In fact, the MHC_1 domain and these mutations were the subject matter of my specific aims and future directions, as I thought it would be beneficial to understand what role this domain plays in the overall function of the protein in humans.
Domain Homology
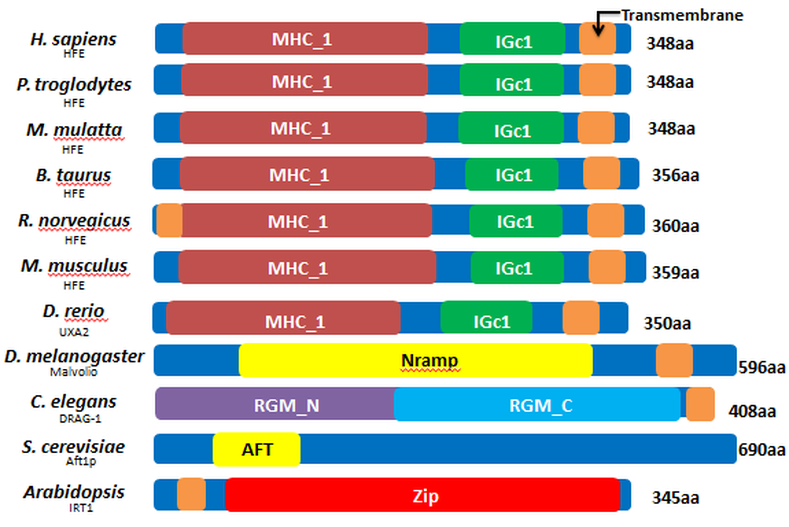
The following image shows the domains of the homologues of the HFE protein. Across the vertebrates the domains are very conserved, indicating that these protein homologues serve very similar functions to the human HFE protein.
Domain Homology Analysis
Three of the invertebrates (C. elegans, D. melanogaster, and Arabidopsis) have different domains from the human HFE protein but still maintains a transmembrane region in some part of the protein. However, S. cerevisiae (yeast) does not share any of domains with human HFE, nor does it have a transmembrane protein. This gives support of why this protein was the least related homologue to the human HFE protein. While the domains of the invertebrates differ greatly from human HFE, these domains likely function in a similar manner to facilitate iron uptake in these species. If they did not, they would likely not be as similar as they are to human HFE.
References
1. EMBL-EBI Training http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
2. Goswami, T., Andrews, N.C. (2006). Hereditary hemochromatosis protein, HFE, interaction with transferrin receptor 2 suggests a molecular mechanism for mammalian iron sensing. Journal of Biological Chemistry, 281: 28494-28498. doi: 10.1074/jbc.C600197200
2. Goswami, T., Andrews, N.C. (2006). Hereditary hemochromatosis protein, HFE, interaction with transferrin receptor 2 suggests a molecular mechanism for mammalian iron sensing. Journal of Biological Chemistry, 281: 28494-28498. doi: 10.1074/jbc.C600197200