Chemical Genetics
What is Chemical Genetics?
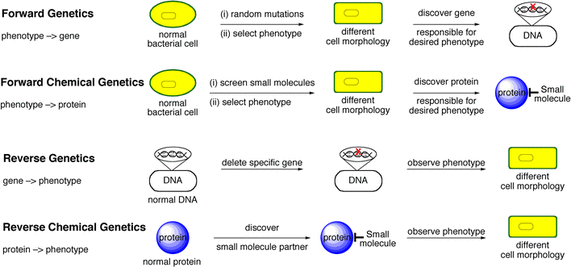 Figure 1: An explanation of chemical genetics in the context of forward and reverse genetics
Figure 1: An explanation of chemical genetics in the context of forward and reverse genetics
Chemical genetics is a method in which small organic molecules are used to disrupt the function of a protein [1]. It can be used in a reverse genetics manor in which the phenotypes that result after the application of a small molecule are studied in order to determine the exact function of a protein of interest. This is similar to the reverse genetic approach in which mutations are induced within a gene to determine which pathways are affected by the now non-functional gene. In the case of chemical genetics, however, the protein itself is affected.
Chemical genetics can also be used in a forward genetics manor where a small molecule is introduced to a cell in order to induce a particular phenotype. Then the protein that interacted with the small molecule to produce such a phenotype is identified. Figure 1 gives a visual explanation of chemical genetics and forward/reverse genetics.
Chemical genetics can also be used in a forward genetics manor where a small molecule is introduced to a cell in order to induce a particular phenotype. Then the protein that interacted with the small molecule to produce such a phenotype is identified. Figure 1 gives a visual explanation of chemical genetics and forward/reverse genetics.
Chemical Genetics Libraries
|
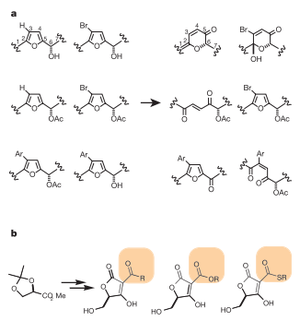
Figure 2: Examples of (a) diversity oriented libraries and (b) focused libraries. Source: [2]
|
There are two types of chemical libraries that can be utilized in chemical genetics: diversity-oriented libraries and focused libraries. Diversity-oriented libraries have a large variety of molecules in order to increase the probability that one of the molecules from the library will affect the protein of interest. Focused libraries are useful after a certain functional group in a molecule as been shown to affect a protein of interest. These libraries focus on that particular functional group in order to optimize the binding of an organic molecule to a protein of interest [2]. Figure 2 gives some examples of these libraries.
In addition to studying protein function, chemical genetics can also be used to identify potential drug candidates that could target a protein of interest. |
Chemical genetics and HFE
|
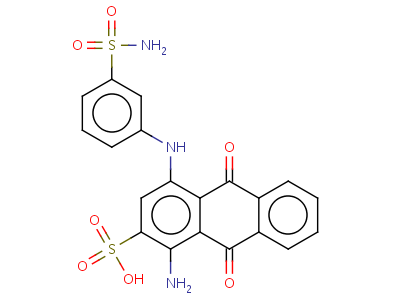
Two databases that can be used to find data on small molecules that may interact with a protein are PubChem and ChemBank. I was not able to find any small molecules that interacted with HFE specifically. However, while looking through the ChemBank database I did find a study performed on the inhibition of iron uptake in mammals (specifically HeLa cells). One of the molecules that was identified to inhibit iron uptake was 1-amino-4-{[3-(aminosulfonyl)phenyl]amino}-9,10-dioxo-9,10-dihydroanthracene-2-sulfonic acid (Figure 2).
|
Figure 2: The structure of 1-amino-4-{[3-(aminosulfonyl)phenyl]amino}-9,10-dioxo-9,10-dihydroanthracene-2-sulfonic acid (click image for source).
|
Analysis
Since this molecule has been shown to inhibit iron uptake in HeLa cells (which are a line of human cells) it is possible that this molecule could interact with HFE. It is also possible that one of the functional groups in this organic molecule would interact with HFE. If this is the case, a focused library with a particular functional group could be created to optimize the binding of an organic molecule with HFE. If this molecule or any similar molecule does not interact with HFE, it is still possible that this kind of molecule could be used as a potential drug to treat hereditary hemochromatosis.
References
1. Brown, J.X., Buckett, P.D., and Wessling-Resnick, M. (2004). Identification of small molecule inhibitors that distinguish between non-transferrin bound iron uptake and transferring-mediated iron transport. Chemical Biology, 11(3), 407-416.
2. Stockwell, B.R. (2004). Exploring biology with small organic molecules. Nature, 432, 846-854. doi:10.1038/nature03196
3. PubChem: https://pubchem.ncbi.nlm.nih.gov/
4. ChemBank: http://chembank.broadinstitute.org/welcome.htm
2. Stockwell, B.R. (2004). Exploring biology with small organic molecules. Nature, 432, 846-854. doi:10.1038/nature03196
3. PubChem: https://pubchem.ncbi.nlm.nih.gov/
4. ChemBank: http://chembank.broadinstitute.org/welcome.htm