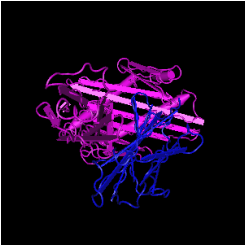
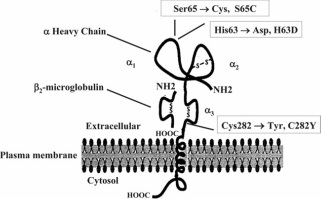
HFE StructureThe human HFE protein is 348 amino acids long. The most common mutation in this protein found in individuals with hereditary hemochromatosis is the C282Y mutation. In this mutation a cytosine at amino acid position 282 is replaced with a tyrosine [1]. This particular mutation causes a break in the disulfide bond in the alpha 3 subunit of the protein [2] (Figure 1). Two other common mutations exist for HH (H63D and S65C), but these mutations do not cause as severe of phenotypes as C282Y [1]. The sequence can be found on the right.
Figure 2: Molecular structure of HFE (click through for source).
|
Figure 1: Diagram of the HFE protein. The Cys282 -> Tyr282 substitution is the cause of the C282Y mutation. Image credit: [6]
HFE Amino Acid SequenceMGPRARPALLLLMLLQTAVLQGRLLRSHSLHYLFMGASEQDLGLSLFEALGY
VDDQLFVFYDHESRRVEPRTPWVSSRISSQMWLQLSQSLKGWDHMFTVDF WTIMENHNHSKESHTLQVILGCEMQEDNSTEGYWKYGYDGQDHLEFCPDTL DWRAAEPRAWPTKLEWERHKIRARQNRAYLERDCPAQLQQLLELGRGVLDQ QVPPLVKVTHHVTSSVTTLRCRALNYYPQNITMKWLKDKQPMDAKEFEPKD VLPNGDGTYQGWITLAVPPGEEQRYTCQVEHPGLDQPLIVIWEPSPSGTLVIG VISGIAVFVVILFIGILFIILRKRQGSRGAMGHYVLAERE |
HFE and Iron Uptake
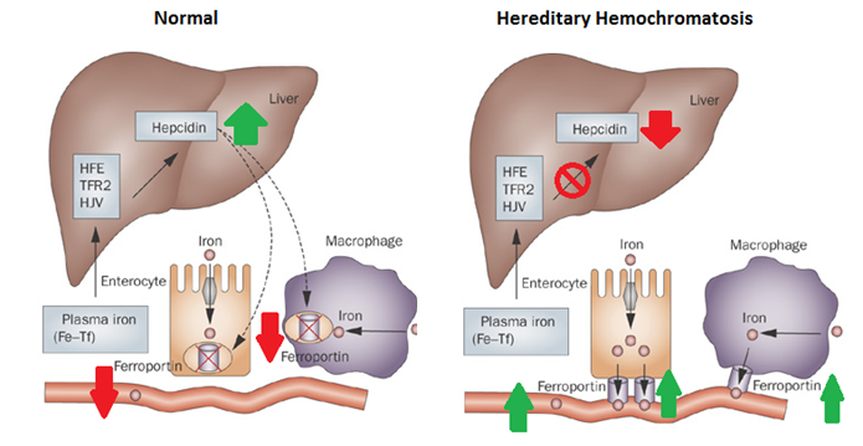
Previous research has shown that HFE mutations affect iron absorption by affecting the production of hepcidin. Hepcidin, which is produced in the liver, is the primary regulator of iron uptake as it controls the presence of ferroporins in the duodenum. When hepcidin levels are low, ferroporins persist and iron is absorbed from the intestine into the bloodstream [3]. When the HFE has a disease-causing mutation hepcidin levels are lowered, allowing for the almost constant uptake of iron [2]. HFE has been shown to affect hepcidin levels via a signal transduction pathway that ultimately results in the transcription (or lack thereof, in the case of HH) of the hepcidin gene [4]. Studies have shown that an interaction of HFE with transferrin receptor 2 (TfR2) is necessary for the progression of the hepcidin pathway [2,5]. The HFE-TfR2-Hepcidin interactions are shown below in figure 3.
Figure 3: The HFE-TFR2-Hepcidin interaction pathway. a) In hereditary hemochromatosis HFE fails to interact with TfR2 and thus the transcription of hepcidin fails. Therefore, when iron levels are high ferroportins persist when hepcidin levels would normally increase and remove them. This causes more iron to flow from the intestines into the bloodstream. b) In a normal individual, when there are high levels of iron the HFE protein interacts with TfR2 to increase the transcription of hepcidin. Hepcidin removes ferroportins, thereby decreasing the amount of iron that flows from the intestine into the bloodstream. Figure adapted from [3]
References
1. Swinkels, D.W., Janssen, M.C., Bergmans, J., Marx, J.J. (2006). Hereditary hemochromatosis: genetic complexity and new diagnostic approaches. Clinical Chemistry, 52(6): 950-68.
2. Gao, J., Chen, J., Kramer, M., Tsukamoto, H., Zhang, A., Enns, C.A. (2009). Interaction of the hereditary hemochromatosis protein HFE with transferrin receptor 2 is required for transferrin-induced hepcidin expression. Cell Metabolism, 9: 217-227. doi: 10.1016/j.cmet.2009.01.010
3. Utzschneider, K.M., Kowdley, K.V., (2010). Hereditary hemochromatosis and diabetes mellitus: implications for clinical practice. Nature Review of Endocrinology, 6, 26-33. doi:10.1038/nrendo.2009.241
4. Swinkles, D.W., Fleming, R.E. (2011). Novel observations in hereditary hemochromatosis: potential implications for clinical strategies. Haematologica, 96(4): 485-488. doi:10.3324/haematol.2011.042036
5. Goswami, T., Andrews, N.C. (2006). Hereditary hemochromatosis protein, HFE, interaction with transferrin receptor 2 suggests a molecular mechanism for mammalian iron sensing. Journal of Biological Chemistry, 281: 28494-28498. doi: 10.1074/jbc.C600197200
6. Bacon, B.R., Adams, P.C., Kowdley, K.V., Powell, L.W., Tavill, A.S. (2011). Diagnosis and management of hemochromatosis: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology, 54(1), 328-43. doi: 10.1002/hep.24330
2. Gao, J., Chen, J., Kramer, M., Tsukamoto, H., Zhang, A., Enns, C.A. (2009). Interaction of the hereditary hemochromatosis protein HFE with transferrin receptor 2 is required for transferrin-induced hepcidin expression. Cell Metabolism, 9: 217-227. doi: 10.1016/j.cmet.2009.01.010
3. Utzschneider, K.M., Kowdley, K.V., (2010). Hereditary hemochromatosis and diabetes mellitus: implications for clinical practice. Nature Review of Endocrinology, 6, 26-33. doi:10.1038/nrendo.2009.241
4. Swinkles, D.W., Fleming, R.E. (2011). Novel observations in hereditary hemochromatosis: potential implications for clinical strategies. Haematologica, 96(4): 485-488. doi:10.3324/haematol.2011.042036
5. Goswami, T., Andrews, N.C. (2006). Hereditary hemochromatosis protein, HFE, interaction with transferrin receptor 2 suggests a molecular mechanism for mammalian iron sensing. Journal of Biological Chemistry, 281: 28494-28498. doi: 10.1074/jbc.C600197200
6. Bacon, B.R., Adams, P.C., Kowdley, K.V., Powell, L.W., Tavill, A.S. (2011). Diagnosis and management of hemochromatosis: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology, 54(1), 328-43. doi: 10.1002/hep.24330